The OncoLux LUMIS™ system enables early diagnosis and targeted treatment intraoperatively to
optimize the potential for curative outcomes.
The OncoLux LUMIS™ system enables early diagnosis and targeted treatment intraoperatively to
optimize the potential for curative outcomes.
The OncoLux LUMIS™ system enables early diagnosis and targeted treatment intraoperatively to
optimize the potential for curative outcomes.
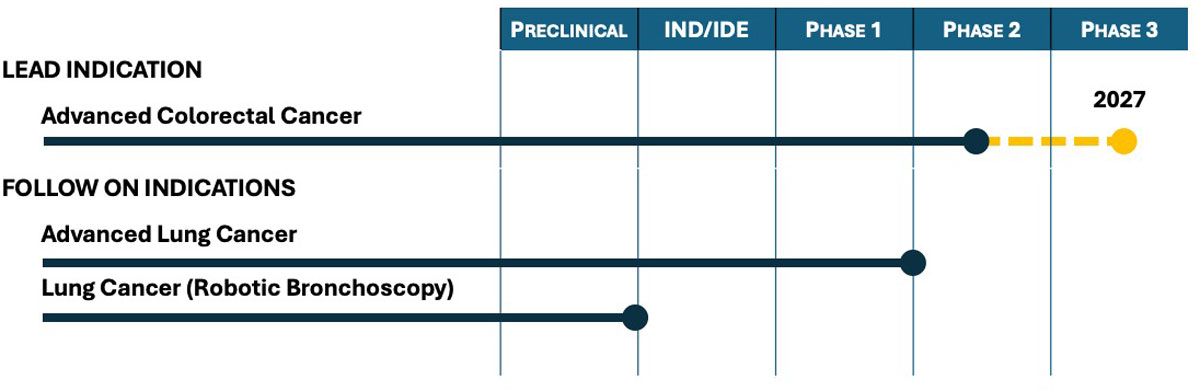
Pipeline
In being compatible with most minimally invasive procedures, OncoLux theranostic technology offers potential clinical use in many cancer sites. The technology has undergone extensive preclinical testing and has been successfully validated clinically in multiple Phase 1 lung and colorectal cancer studies. The company’s lead indication has now advanced into a Phase 2 study.
Colorectal Cancer Indication
Patients with locally advanced T4 or recurrent tumors undergoing surgery.
Study Attributes
“See and Treat” during robotic-assisted laparoscopic procedure to reduce rate of disease recurrence
Use of 5-ALA (Gleolan®) enables:
– Florescence Guided Surgery and
– PDTNo systemic toxicities or side effects, minimal damage to healthy tissue
PDT System IDE approved; provides precise, repeatable administration of PDT
Benefits of intraoperative adjuvant therapy (such as IORT) but fully compatible with MIS/RAS procedures
Study Schedule
| Study Status | Recruiting |
| Study Start | June 2024 |
| Phase 1 Primary Completion | December 2024 |
| Phase 2 Start | February 2025 |
| Phase 2 Primary Completion | December 2026 (estimated) |
| Preliminary Study Efficacy Readout | May 2026 (estimated) |
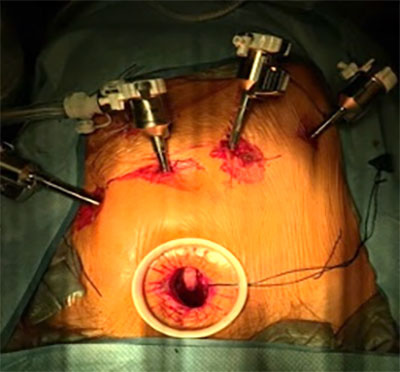
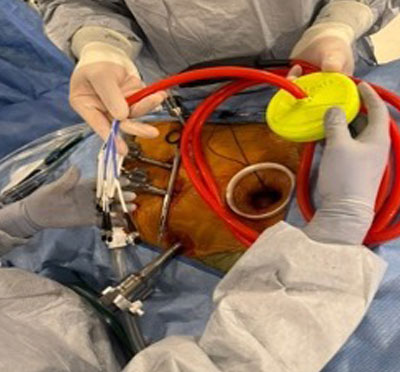

F Jehan, MD, S McIntyre, MD, E Gawrys, PhD, G Shafirstein, PhD, A Dakwar, MD, FACS, Roswell Park Comprehensive Cancer Center, Buffalo, NY. ACS Clinical Congress 2025

Colorectal Tumor Biopsy Specimen demonstrating PpIX Fluorescence
OncoLux’s Aurora™ imaging system has been installed at the Roswell Park Cancer Center clinical site for use in the Phase 2 colorectal study (NCT06307548) as a benchtop ex-vivo imaging system to confirm 5-ALA/PpIX fluorescence. This system provides clinical investigators a valuable high-sensitivity tool to better detect and classify tissue specimens.
Lung Cancer Indication
Patients with suspicious peripheral lung nodules after screening (low-dose CT) that are undergoing biopsy via robotic bronchoscopy
LUMIS™ offers a theranostic capability to current robotic bronchoscopy tissue biopsy systems to perform “optical biopsy” and the option to treat the lesion in-situ during the procedure
Theranostic fiber optic probe run in the working channel of the robotic catheter
Intraoperative “optical biopsy” using proprietary optical fluorescence disease detection
Optional intraoperative therapy immediately after diagnosis for “see and treat” during robotic bronchoscopy procedure
Early diagnosis and highly targeted delivery of therapy can expedite patient time treatment time and improve potential for curative outcomes

The OncoLux LUMIS™ system will enable earlier diagnosis and highly targeted delivery of real-time ‘In-Situ’ therapies that minimize patient time from Dx to Tx thus optimizing the potential for curative outcomes.
